Development of broadly neutralizing antibodies against SARS-CoV-2 using improved mRNA-lipid nanoparticle immunization technology.
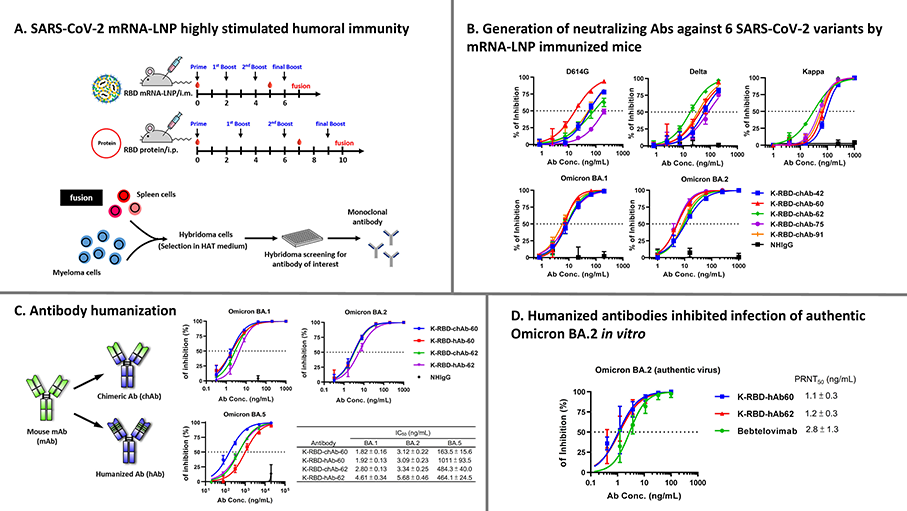
To help mitigate the risks of COVID-19 to human health, scientists have been continually working to develop and improve many types of prophylactic and therapeutic drugs. In addition to vaccines, monoclonal antibodies (mAbs) that bind to and neutralize SARS-CoV-2 virus have been applied as complementary and alternative treatments. In our studies, we employed a classical hybridoma fusion strategy coupled with state-of-the-art mRNA-lipid nanoparticle (LNP) immunization to create mAbs for SARS-CoV-2. This time-saving approach can circumvent common biological and technical hurdles, such as difficulties in expressing membrane proteins, antigen instability, and the lack of post-translational modifications on recombinant antigens. In particular, we generated mAbs against the receptor binding domain (RBD) of SARS-CoV-2 spike (S) protein. Compared with traditional protein-based immunization approaches, inoculation of mice with RBD mRNA-LNP induced higher titers of serum antibodies and markedly increased serum neutralizing activity. This method allowed us to successfully generate a set of K-RBD-mAbs targeting the Omicron variant, five of which show strong binding and neutralizing activities toward all SARS-CoV-2 variants of concern (Alpha, Beta, Gamma, Delta and Omicron). We then further performed antibody humanization on broadly neutralizing chimeric antibodies to create K-RBD-hAb-60 and -62, which retain excellent neutralizing potency against Omicron BA.1 and BA.2. In addition to yielding mAbs that may be used to combat SARS-CoV-2, our work generally demonstrates that the mRNA-LNP immunization platform may be useful for quickly generating highly functional mAbs against emerging infectious diseases. Two reports on this project have been published in the International Journal of Pharmaceutics and Heliyon (Cell Press); the lead authors of the studies are Dr. Fu-Fei Hsu and Dr. Ruei-Min Lu, respectively.
Paper link:
(1) https://www.sciencedirect.com/science/article/pii/S0378517322008110?via%3Dihub
(2) https://www.sciencedirect.com/science/article/pii/S2405844023027949?via%3Dihub