A critical overview of current progress for COVID-19: development of vaccines, antiviral drugs, and therapeutic antibodies
- Author:Monika Kumari, Ruei-Min Lu, Mu-Chun Li, Jhih-Liang Huang, Fu-Fei Hsu, Shih-Han Ko, Feng-Yi Ke, Shih-Chieh Su, Kang-Hao Liang, Joyce Pei-Yi Yuan, Hsiao-Ling Chiang, Cheng-Pu Sun, I-Jung Lee, Wen-Shan Li, Hsing-Pang Hsieh, Mi-Hua Tao, and Han-Chung Wu*
- Journal: Journal of Biomedical Science (2022) 29:68 https://jbiomedsci.biomedcentral.com/articles/10.1186/s12929-022-00852-9
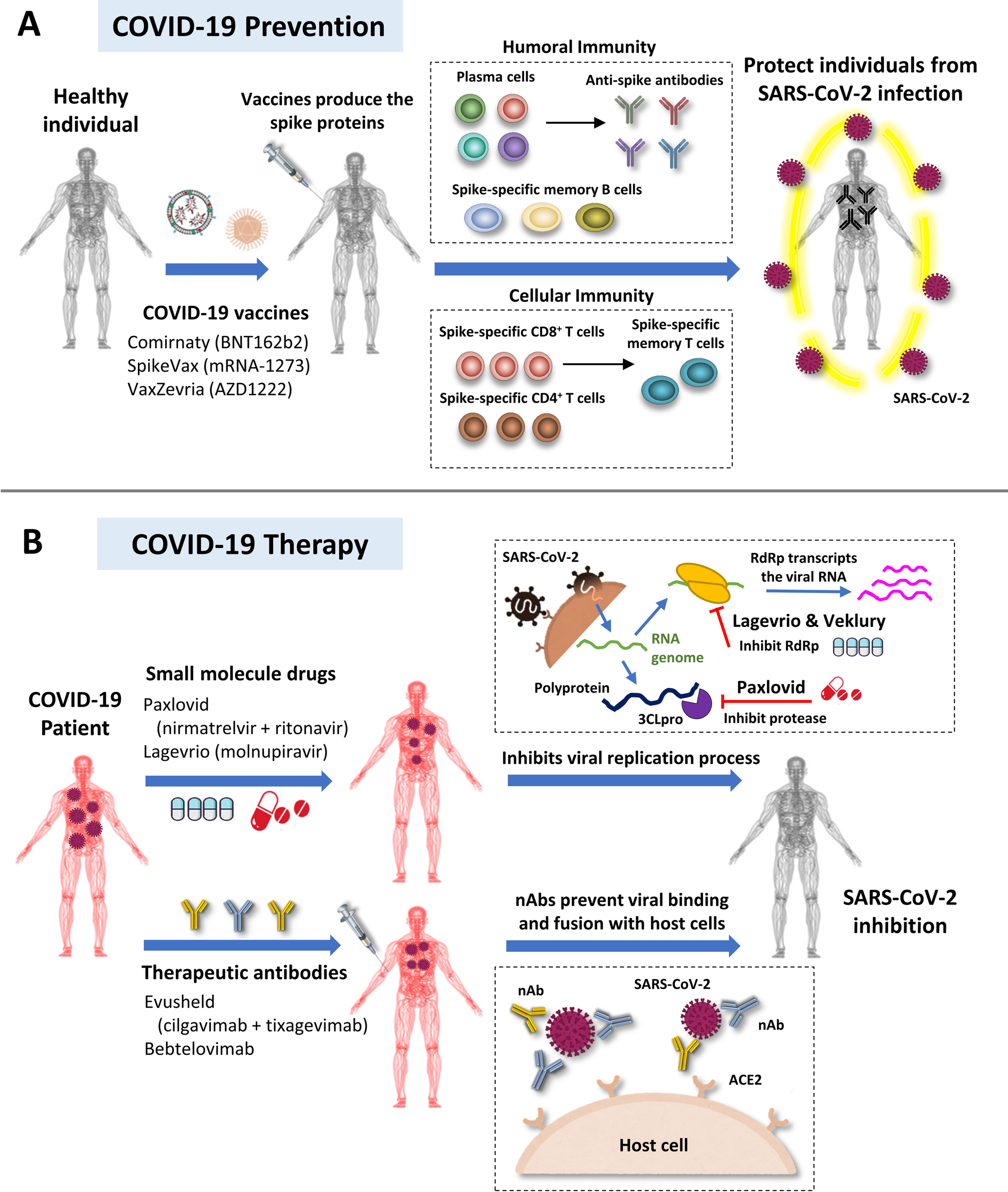
The novel coronavirus disease (COVID-19) pandemic remains a global public health crisis, presenting a broad range of challenges. To help address some of the main problems, the scientific community has designed vaccines, diagnostic tools, and therapeutics for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. Outbreaks of more infectious variants continue to diminish the utility of available vaccines and drugs. Thus, the effectiveness of vaccines and drugs against the most current variants is a primary consideration in the continual analyses of clinical data that supports updated regulatory decisions. The first two vaccines granted Emergency Use Authorizations (EUAs), BNT162b2 and mRNA-1273, still show more than 60% protection efficacy against the most widespread current SARS-CoV-2 variant, Omicron. This variant carries more than 30 mutations in the spike protein, which has largely abrogated the neutralizing effects of therapeutic antibodies. Fortunately, some neutralizing antibodies and antiviral COVID-19 drug treatments have shown continued clinical benefits.
In this review article, Dr. Han-Chung Wu lead his research team at ICOB and BioTReC have provided a framework to understand the ongoing development efforts for different types of vaccines and therapeutics, including small molecule and antibody drugs. Dr. Wu’s group with the help of Dr. Wen-Shan Li’s group has discussed the structure-based framework for lipid nanoparticles (LNPs) designed to deliver mRNA. Moreover, Dr. Hsing-Pang Hsieh’s and Dr. Mi-Hua Tao’s teams provide the newest information about small molecule drugs and viral vector-based vaccines against SARS-CoV-2 infection. Finally, this article aims to highlights the ripple effects of newly emergent variants on different strategies that have been used for COVID-19 prevention and therapy. This information can be an essential reference for government, academia, and industries to overview the SARS-CoV-2 pandemic and develop the vaccines and anti-viral drugs.
First authors: Dr. Monika Kumari (postdoctoral scholar) at ICOB and Dr. Ruei-Min Lu (assistant research scientist) at BioTReC