ENO1 Promotes Lung Cancer Metastasis via HGFR and WNT Signaling–Driven Epithelial-to-Mesenchymal Transition
- 作者:Hsin-Jung Li, Feng-Yi Ke, Chia-Ching Lin, Mei-Yi Lu, Yi-Huei Kuo, Yi-Ping Wang, Kang-Hao Liang, Shin
- 期刊: Cancer Research Aug. 2021 Vol. 81, Issue 15 https://cancerres.aacrjournals.org/content/81/15/4094
Neutralizing ENO1 prevents lung cancer cell growth and metastasis
For many types of cancer, the cell-surface molecule ENO1 (α-enolase) is considered to be a promising target for new diagnostic tools and therapeutics. A recent study by Dr. Han-Chung Wu’s group at the Institute of Cellular and Organismic Biology, Academia Sinica, has greatly advanced our knowledge of how ENO1 promotes tumorigenesis and introduces a new tool for preventing cancer growth and metastasis. The study was published in Cancer Research [1].
In their previous study, Wu’s group targeted ENO1 with a peptide called pHCT74. The authors incorporated pHCT74 into an innovative targeted drug delivery system that combines potent small molecule drugs with highly specific protein-targeting agents. This system leverages the benefits of the two therapeutic strategies and eliminates some of their key disadvantages, such as off-target toxicity. By increasing the therapeutic efficacies of small molecule drugs and reducing their side effects, the system can be used to improve treatments for many cancers in a previous study published in the top-ranking journal, Science Translational Medicine [2]. ENO1 is best known as a metabolic enzyme involved in the synthesis of pyruvate, but the protein is also highly expressed on the surface of cells in most tumors. This elevated expression of ENO1 in cancer cells is correlated with reduced survival rate and poor prognosis in many cancers. Wu’s group has studied the role of ENO1 in cancer and its potential as a therapeutic target for several years. Recently, Wu’s group showed that ENO1 activates two signaling pathways to stabilize the tumorigenic factor, SLUG, which then promotes epithelial-mesenchymal transition and invasion/metastasis in lung cancer. In this study, they further examined the biological role of ENO1 in tumor progression and invasion, demonstrating that ENO1-promoted lung cell invasion, tumor metastasis and progression specifically involves the HGFR-Src-PI3K-AKT-GSK3β-SLUG and HGFR-WNT-GSK3β-SLUG signaling axes. This mechanism of action suggests that in addition to its utility as a biomarker, ENO1 may also be a valuable target for lung cancer therapeutics.
In order to test the potential of ENO1 as a therapeutic target, the authors developed a neutralizing antibody, called chENO1-22, which is able to prevent ENO1 signaling. Application of chENO1-22 to cancer cells was found to decrease the level of SLUG, thereby reducing lung cancer cell growth and invasion. Importantly, this neutralizing antibody was also able to improve survival in a lung cancer xenograft mouse model, and it may be therefore be considered a promising therapeutic candidate for lung cancer. A US Provisional Application for Patent has been filed on the technology, as it is expected to be useful as a research tool and potentially as a clinically approved therapeutic in the future.
The current research paper, “ENO1 promotes lung cancer metastasis via HGFR and WNT signaling-driven epithelial-mesenchymal transition,” will be officially published on Aug 2nd, 2021. The full text of the paper: https://cancerres.aacrjournals.org/content/early/2021/06/17/0008-5472.CAN-20-3543. The first author of this paper is Dr. Hsin-Jung Li from the Institute of Cellular and Organismic Biology, Academia Sinica. The research team includes Distinguished Researcher Han-Chung Wu of the Institute of Cellular and Organismic Biology, Academia Sinica and Academician Pan-Chyr Yang of the Institute of Biomedical Sciences, Academia Sinica.
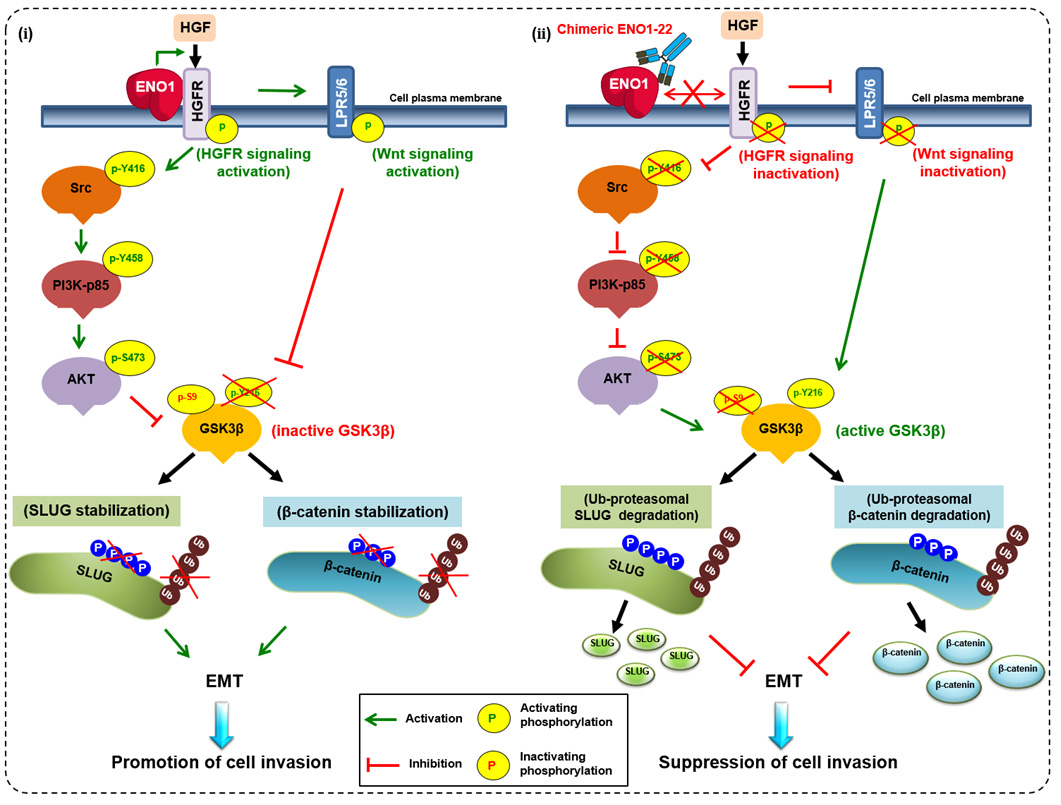
The studies by Dr. Han-Chung Wu and his group on the cellular mechanisms of ENO1-mediated tumorigenesis and the development of neutralizing antibodies were published in top journals. A working model depicts ENO1 promotion of cell invasion through regulation of the HGFR-Src-PI3K-AKT-GSK3β-SLUG and HGFR-WNT-GSK3β-SLUG axes. (ⅰ) ENO1 increases the phosphorylation of HGFR and Wnt co-receptor LPR5/6 and decreases GSK-3β activity to enhance SLUG stabilization and suppress E-cadherin expression. Ultimately, cancer cell invasion capacity is enhanced. (ⅱ) The chENO1-22 monoclonal antibody inhibits cancer cell invasion by blocking ENO1-HGFR axis-mediated downstream signaling to promote SLUG protein ubiquitination and degradation. Based on these discoveries and innovations, Dr. Wu’s group is poised to create new and effective treatments for cancer.
肺癌惡化機制大發現!新研發抗體大幅降低癌症形成及轉移
在癌症研究中,α-烯醇酶 (α-enolase, 又稱ENO1) 被發現高量表達於癌細胞。近期,中央研究院細胞與個體生物學研究所特聘研究員兼任生醫轉譯研究中心主任吳漢忠的研究團隊發表了一項研究,證實ENO1也存在癌細胞的細胞膜上,並且掌握了ENO1如何促進肺癌細胞生長與轉移,以及如何阻斷這項機制。研究成果已發表在國際期刊《癌症研究》(Cancer Research)[1]。
本實驗室之前已成功地篩選專一性結合腸癌之新穎標的胜肽pHCT74,並且利用此胜肽成功開發標的微脂體,此標的微脂體明顯抑制腫瘤的生長及大幅提升其存活率,並且成功鑑定出pHCT74胜肽分子靶標蛋白是 ENO1。ENO1 是一種參與丙酮酸合成的代謝酶,已知在大多數腫瘤的細胞表面會大量表達,此現象與許多癌症的存活率降低和預後不良有著顯著的關聯性,相關研究成果已發表於國際期刊《科學轉譯醫學》(Science Translational Medicine)[2]。近期,研究團隊進一步發現,它會雙重活化重要的致癌訊息傳遞,包括肝細胞生長因子受體 (HGFR) 與Wnt 信號,進而穩定促癌轉移分子SLUG及β-catenin的蛋白,使得癌細胞獲得強大的侵襲及轉移的能力,最終導致腫瘤轉移至身體各處!
為了能精準辨識出癌細胞膜上的ENO1,研究團隊研發出相對應的中和性單株抗體chENO1-22,可抑制ENO1的訊息傳遞功能、降低肺癌細胞中SLUG及β-catenin蛋白的表現,進而降低癌細胞的生長和轉移的能力。在肺癌的動物實驗中,chENO1-22展現了抑制腫瘤轉移能力及提升小鼠的存活率;此外,在肺癌的動物實驗,同時使用chENO1-22 抗體和肺癌臨床用藥-HGFR小分子抑制劑 (Crizotinib) 的「聯合療法」,可進一步增加抑制腫瘤轉移能力及提升小鼠的存活率。這些研究發現,更深入剖析ENO1促使癌症惡化的分子機制,並為轉移性癌症治療引入了具潛力的治療性抗體。
吳漢忠表示:「ENO1可見於許多種癌細胞,如:肺癌、攝護腺癌、肝癌等,因此,這項研究成果有潛力廣泛應用在各種不同的癌症診斷和治療。」目前,此中和性抗體chENO1-22在癌症的診斷及標靶治療的應用,深具發展潛力,已申請美國專利,未來可望化研為用,造福人群。
本篇研究論文:ENO1 promotes lung cancer metastasis via HGFR and WNT signaling-driven epithelial-mesenchymal transition於2021年08月02日正式發表。論文全文:https://cancerres.aacrjournals.org/content/early/2021/06/17/0008-5472.CAN-20-3543。此論文第一作者為中研院細胞與個體生物學研究所李欣蓉博士。研究團隊包含中研院細胞與個體生物學研究所吳漢忠特聘研究員及生物醫學研究所楊泮池院士。
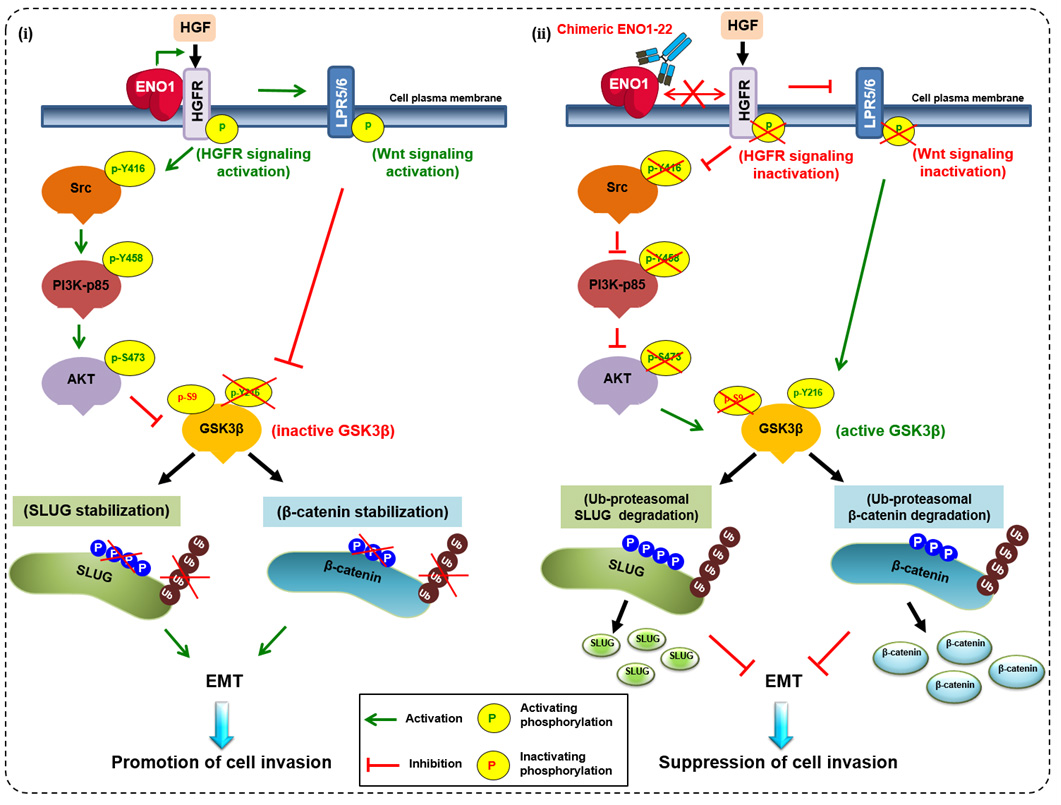
圖說:研究團隊發現ENO1 增加 HGFR 和 Wnt 共受體 LPR5/6 的磷酸化並降低 GSK-3β 活性,以增強 SLUG 穩定性並抑制 E-cadherin蛋白表達; 最終增強癌細胞侵襲能力。在利用 chENO1-22 抗體通過阻斷 ENO1-HGFR-Wnt 軸介導的下游信號傳導,以促進 SLUG 蛋白泛素化和降解來抑制癌細胞侵襲。
References:
- Li, H.J., et al., ENO1 promotes lung cancer metastasis via HGFR and WNT signaling-driven epithelial-mesenchymal transition. Cancer Res, 2021.
- Wu, C.H., et al., alpha-Enolase-binding peptide enhances drug delivery efficiency and therapeutic efficacy against colorectal cancer. Sci Transl Med, 2015. 7(290): p. 290ra91.